In highly regulated industries, Device Lab Management is not just about organizing devices. It directly impacts compliance, data protection, and day-to-day operations. Organizations working with sensitive data, such as healthcare providers, financial institutions, and pharmaceutical companies, need a structured and secure approach to managing their device labs. Without it, the risks range from compliance penalties to data exposure and operational delays.
A well-managed device lab supports consistent testing, protects sensitive information, and keeps teams aligned with regulatory requirements. Below are practical best practices that help maintain a secure and reliable device lab environment.
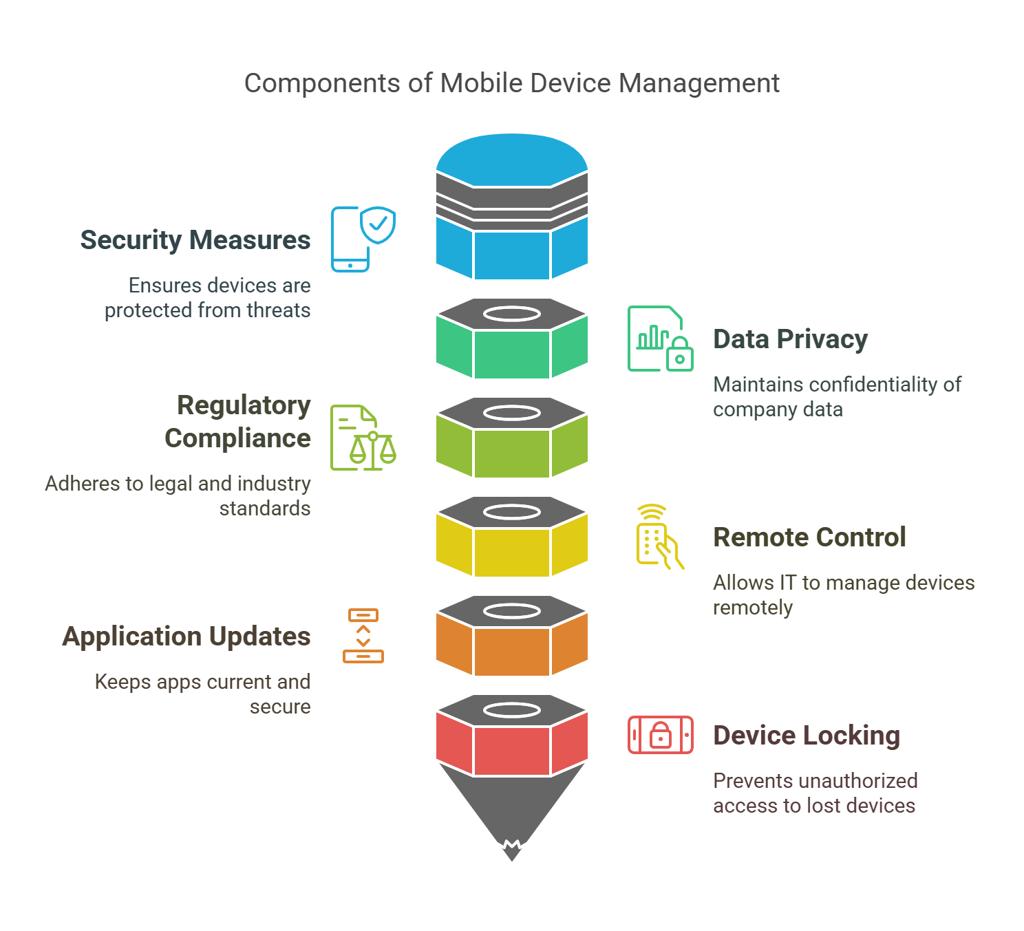
1. Implement Strict Access Controls
Controlling access is the foundation of secure Device Lab Management. Not everyone should have the same level of access to devices or configurations.
Start by applying role-based access control so each user only has permissions relevant to their role. Add multi-factor authentication to strengthen login security, especially for remote access scenarios. It is also important to review access logs regularly to spot unusual activity or unauthorized attempts.
Platforms like Kobiton make it easier to manage controlled access to real devices while maintaining visibility into user activity. This kind of structured access control helps meet compliance standards such as HIPAA and GDPR while reducing internal risks.
2. Maintain Device Inventory and Tracking
A clear and up-to-date inventory is essential for managing any device lab, especially in regulated environments. Every device should be accounted for and documented with key details such as operating system version, installed applications, ownership, and usage history.
Using automated tracking systems saves time and reduces human error. It also helps teams quickly locate devices, plan maintenance, and monitor device status. When devices are properly tracked, it becomes much easier to prevent unauthorized usage and maintain a controlled testing environment.
Strong inventory management also supports audit readiness, which is often a requirement in regulated industries.
3. Keep Devices Updated with Security Patches
Outdated devices are one of the most common entry points for security issues. Regular updates are necessary to fix known vulnerabilities and maintain system stability.
Set a consistent schedule for applying operating system updates, security patches, and firmware upgrades. Where possible, use automation to handle updates without interrupting active testing workflows. This keeps devices secure without slowing down your team.
A platform like Kobiton can help maintain device consistency across environments, making it easier to keep devices aligned with required security standards.
4. Secure Data Storage and Handling
Device labs often process or store sensitive data, which means data protection must be handled carefully at every stage.
All data should be encrypted both when stored and when transmitted. Access to data should be limited based on user roles, and logs should be maintained for all data related activities. When a device is retired or reassigned, it must go through a secure data wiping process to prevent any residual data exposure.
Following strict data handling practices not only protects user information but also supports compliance requirements and builds trust with stakeholders.
5. Monitor and Audit Device Usage
Ongoing monitoring gives visibility into how devices are being used and helps identify unusual patterns early. This is especially important in regulated industries where misuse or unauthorized activity can have serious consequences.
Set up centralized logging and real time monitoring to track device activity. Regular audits should be part of your routine to confirm that policies are being followed and that no gaps have been introduced over time.
Consistent monitoring also improves operational efficiency by highlighting underused devices or bottlenecks in testing workflows.
6. Train Staff on Security Protocols
Technology alone is not enough to maintain a secure device lab. The people using the lab need to understand the processes and their responsibilities.
Regular training sessions should cover topics such as secure device handling, access policies, data protection practices, and compliance requirements. Make training practical and scenario based so teams can apply what they learn in real situations.
Human error is still one of the leading causes of security incidents. Well informed staff significantly reduce that risk.
Conclusion
Secure Device Lab Management is a core requirement for organizations operating in regulated industries. It affects compliance, data protection, and the reliability of testing processes.
By putting strong access controls in place, maintaining accurate device tracking, keeping systems updated, securing data, monitoring usage, and training staff, organizations can reduce risk and maintain a stable, compliant environment.
A structured approach to Device Lab Management, supported by reliable platforms like Kobiton, helps teams stay prepared for audits while keeping operations running smoothly.
